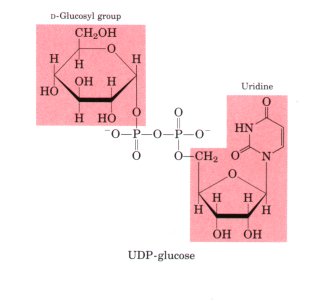
- In a wide range of organisms, excess glucose is converted into polymeric forms for storage and transport. The principal storage forms of glucose are glycogen in vertebrates and many microorganisms, and starch in plants.Many of the reactions in which hexoses are transformed or polymerized involve a different type of activating group, a nucleotide bound to the anomeric hydroxyl of the sugar through a phosphate ester linkage, Sugar Nucleotide.
- Sugar nucleotides are the substrates for polymerization into disaccharides, glycogen, starch, cellulose, and more complex extracellular polysaccharides.
- The role of sugar nucleotides (specificallyUDP-glucose) in the biosynthesis of glycogen and many other carbohydrate derivatives was discovered by Luis Leloir.
The suitability of sugar nucleotides for biosynthetic reactions stems from several properties:
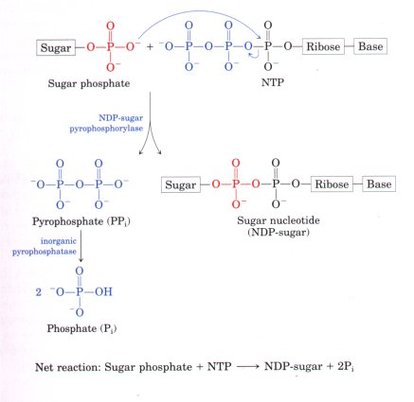
1. Their formation by the condensation of a nucleoside triphosphate with a hexose phosphate splits one high-energy bond and releases PPi, which is further hydrolyzed by inorganic pyrophosphatase; there is a net cleavage of two high-energy bonds. The resulting large, negative, free-energy change drives the synthetic reaction and reflects a strategy common to many biological polymerization reactions.
1. Their formation by the condensation of a nucleoside triphosphate with a hexose phosphate splits one high-energy bond and releases PPi, which is further hydrolyzed by inorganic pyrophosphatase; there is a net cleavage of two high-energy bonds. The resulting large, negative, free-energy change drives the synthetic reaction and reflects a strategy common to many biological polymerization reactions.
2. Although the chemical transformations of sugar nucleotides do not involve the atoms of the nucleotide itself, the sugar nucleotide molecule offers many potential groups for noncovalent interactions with enzymes; the free energy of binding contributes significantly to the catalytic activity of the enzyme.
3. Like phosphate, the nucleotidyl group is an excellent leaving group, activating the sugar carbon to which it is attached so as to facilitate nucleophilic attack.
4. By "tagging" some hexoses with nucleotidyl groups, cells may set them aside for one purpose (glycogen synthesis, for example) in a pool separate from hexose phosphates earmarked for another purpose (such as glycolysis).
3. Like phosphate, the nucleotidyl group is an excellent leaving group, activating the sugar carbon to which it is attached so as to facilitate nucleophilic attack.
4. By "tagging" some hexoses with nucleotidyl groups, cells may set them aside for one purpose (glycogen synthesis, for example) in a pool separate from hexose phosphates earmarked for another purpose (such as glycolysis).
UDP Glucose is the substrate for Glycogen Synthesis
D-Glucose + ATP ---------------> D-glucose-6-phosphate + ADP
Glucose-6-phosphate <----------------------> glucose-1-phosphate
Glucose-1-phosphate + UTP <----------------------------> UDP-glucose + PPi
- In animals and some microorganisms, excess glucose available from carbohydrates in the diet or from gluconeogenesis is stored as glycogen.
- The starting point for synthesis of glycogen is glucose-6-phosphate. This can be derived from free glucose by the hexokinase reaction:
D-Glucose + ATP ---------------> D-glucose-6-phosphate + ADP
- To initiate glycogen synthesis, the glucose-6-phosphate is reversibly converted into glucose-1-phosphate by phosphoglucomutase:
Glucose-6-phosphate <----------------------> glucose-1-phosphate
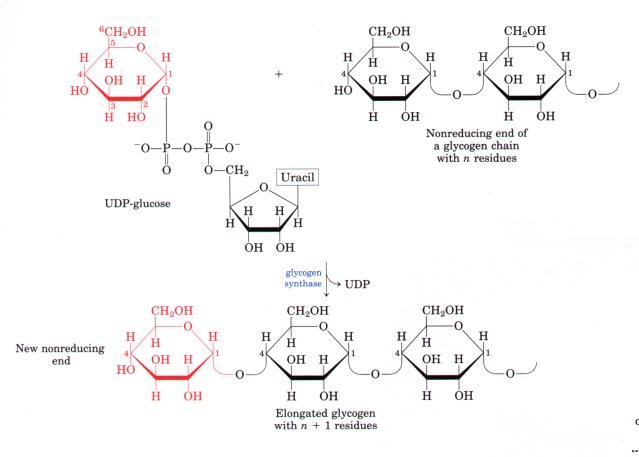
- The formation of UDP-glucose by the action of UDP-glucose pyrophosphorylase is a key reaction in glycogen biosynthesis:
Glucose-1-phosphate + UTP <----------------------------> UDP-glucose + PPi
- This reaction proceeds in the direction of UDP-glucose formation because pyrophosphate is rapidly hydrolyzed to orthophosphate by inorganic pyrophosphatase (ΔG°' = -25 kJ/mol).
- UDP-glucose is the immediate donor of glucose residues in the enzymatic formation of glycogen by the action of glycogen synthase, which promotes the transfer of the glucosyl residue from UDP-glucose to a nonreducing end of the branched glycogen molecule.
- The overall equilibrium of the path from glucose-6-phosphate to lengthened glycogen greatly favors synthesis of glycogen.
- Glycogen synthase requires as a primer an (α1→4) polyglucose chain or branch having at least four glucose residues.
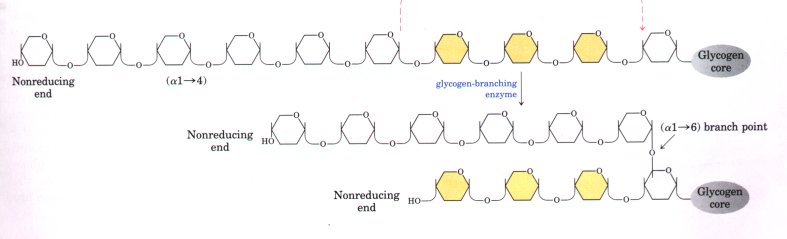
- Glycogen synthase cannot make the (α1→6) bonds found at the branch points of glycogen, instead, these are formed by a glycogen-branching enzyme, amylo (1→4) to (1→6) transglycosylase or glycosyl-(4→6)-transferase.
- Glycosyl-(4→6)-transferase catalyzes transfer of a terminal fragment of six or seven glucosyl residues from the nonreducing end of a glycogen branch having at least eleven residues to the C-6 hydroxyl group of a glucose residue of the same or another glycogen chain at a more interior point, thus creating a new branch.
- Further glucosyl residues may be added to the new branch by glycogen synthase. The biological effect of branching is to make the glycogen molecule more soluble and to increase the number of nonreducing ends, thus making the glycogen more reactive to both glycogen phosphorylase and glycogen synthase.
Glycogenin Primes the Initial Sugar Residues
in Glycogen
· Glycogen synthase cannot initiate a new glycogen chain de novo. It requires a primer, usually a preformed α(1-4) polyglucose chain or branch having at least eight glucose residues.
· How is a new glycogen molecule initiated? The intriguing protein glycogenin is both the primer on which new chains are assembled and the enzyme that catalyzes their assembly.
· The first step in the synthesis of a new glycogen molecule is the transfer of a glucose residue from UDP-glucose to the hydroxyl group of Tyr194 of glycogenin, catalyzed by the protein’s intrinsic glucosyltransferase activity.
· The nascent chain is extended by the sequential addition of seven more glucose residues, each derived from UDP-glucose; the reactions are catalyzed by the chain-extending activity of glycogenin.
· At this point, glycogen synthase takes over, further extending the glycogen chain.
· Glycogenin remains buried within the particle, covalently attached to the single reducing end of the glycogen molecule
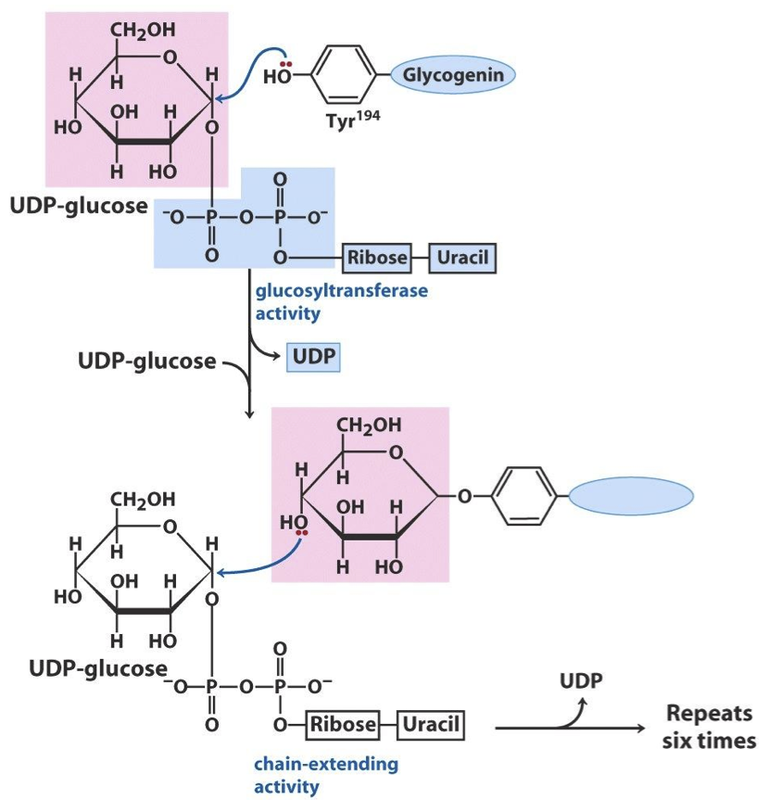
MECHANISM : Glycogenin catalyzes two distinct reactions. Initial attack by the
hydroxyl group of Tyr194 on C-1 of the glucosyl moiety of UDP-glucose results
in a glucosylated Tyr residue. The C-1 of another UDP-glucose molecule is
now attacked by the C-4 hydroxyl group of the terminal glucose, and this
sequence repeats to form a nascent glycogen molecule of eight glucose
residues attached by α(1-4) glycosidic linkages