Mitochondrial Electron Transport Chain:Electrons from NADH and FADH2 are transported to the ultimate electron acceptor, O2 via various protein-bound redox centers present in inner mitochondrial membrane. The free energy released is used to pump proton across the membrane, and the energy of proton gradient thus build is used to drive synthesis of ATP by F0, F1 ATPase (or ATP synthase) system.
Mitochondria
In most aerobic organisms, mitochondria is the major site for the reactions of PDC, citric acid cycle, and ATP generation through electron transport chain and oxidative phosphorylation. Hence called powerhouse of cells.
•It is an intracellular organelle with a size as big as a bacterium.
•It has two membranes outer and inner mitochondrial membranes.
•Mitochondria has its own DNA encoding some of the genes required in ETC and other functions.
Mitochondria also have their own transcription and translation machinery. The mitochondrial ribosomes similar to that of bacteria.
Membrane-Bound Electron Carriers
Mitochondria
In most aerobic organisms, mitochondria is the major site for the reactions of PDC, citric acid cycle, and ATP generation through electron transport chain and oxidative phosphorylation. Hence called powerhouse of cells.
•It is an intracellular organelle with a size as big as a bacterium.
•It has two membranes outer and inner mitochondrial membranes.
•Mitochondria has its own DNA encoding some of the genes required in ETC and other functions.
Mitochondria also have their own transcription and translation machinery. The mitochondrial ribosomes similar to that of bacteria.
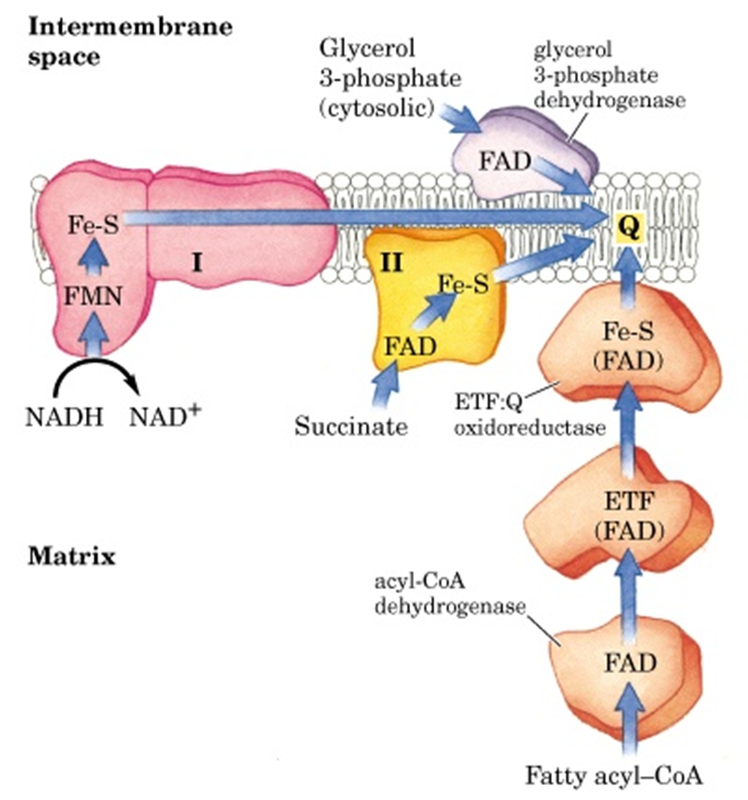
- Virtually all the complexes of electron transport chain and oxidative phosphorylation are located in the inner mitochondrial membrane.
- Electrons from NADH and FADH2 are transported to the ultimate electron acceptor, O2 via four protein complexes containing various redox centers, called complex I, II, III and IV.
- Electrons are transferred through different electron-carriers sequentially in mitochondrial membrane. Most of electron-carriers (except coenzyme Q) are proteins with prosthetic groups capable of accepting or donating electrons.
Membrane-Bound Electron Carriers
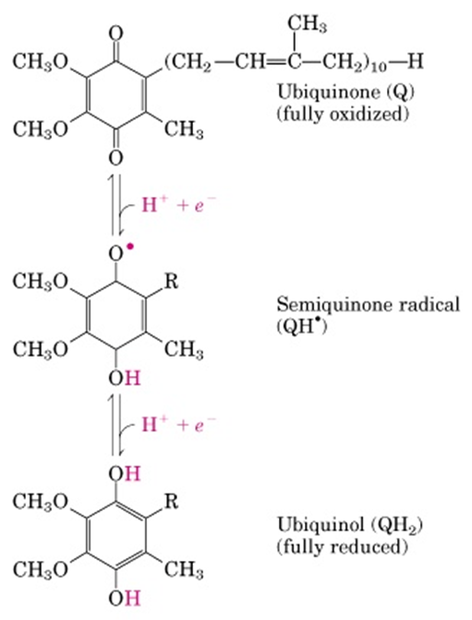
Ubiquinone or Coenzyme Q
- It is a highly hydrophobic, lipid soluble benzoquinone with along isoprenoid side chain.
- Ubiquinone can accept one electron to become semiubiquinone radical or it can accept two electron to become ubiquinol.
- Due to its small size and lipid solubility, it can freely diffuse through lipid bilayer, thus it is capable of shuttling the reducing equivalents (electron) among other less mobile electron carriers.
- Since it carries both electron and protons, it plays central role in coupling electron flow to proton movement.
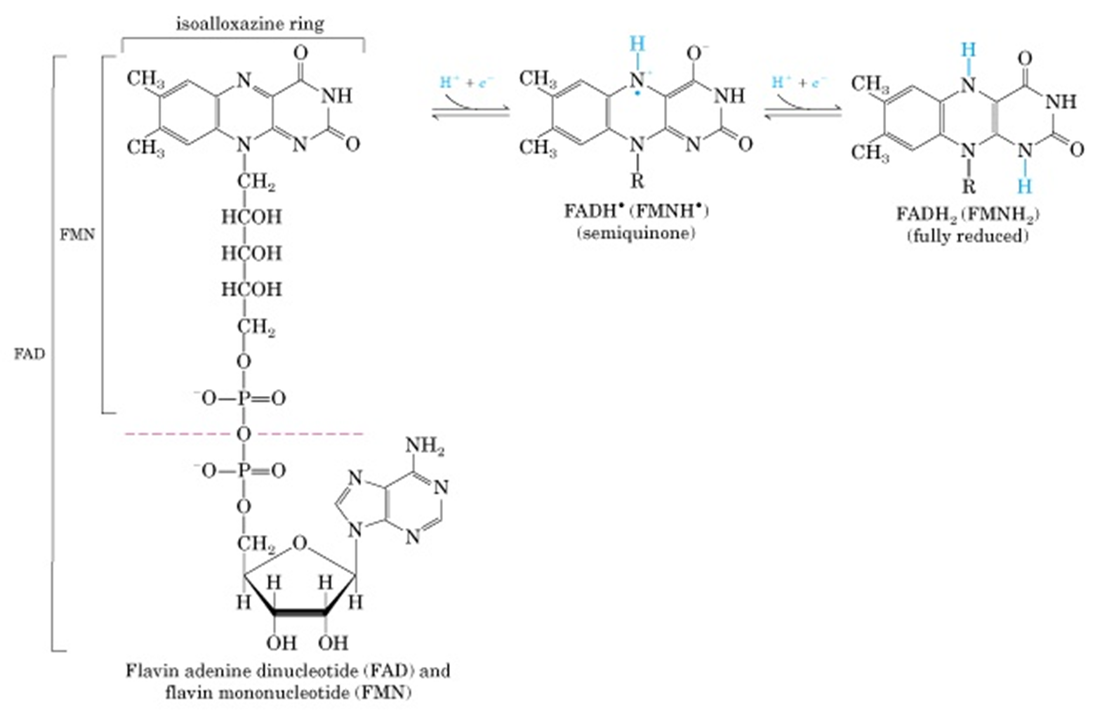
FLAVOPROTIEN:
- Proteins tightly (or covalantly) bound to Flavin nucleotides FMN or FAD.
- The reduction potential of flavin nucleotides depends on the protein it is bound to. Local interaction of functional groups of amino acids distarts the electron orbitals of flavin ring, this changing the stability of oxidized or reduced forms.
- Flavoproteins are capable of accepting or donating one or two electrons.
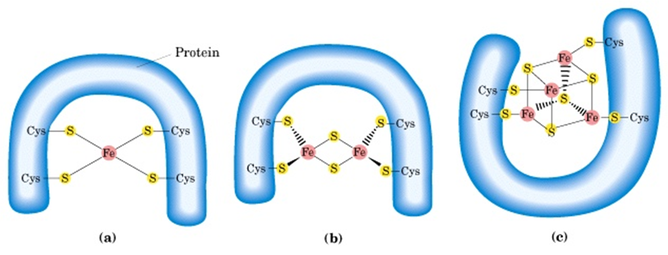
Iron sulfur proteins
- These are proteins containing Fe atom co-ordidated to sulfur atoms of either Cys residues of the proteins or also with inorganic sulfur atoms. The Fe atom is oxidized or reduced
- The reduction potential of Fe-S proteins varies from -0.65 to +0.45V depending on the microenvironment around Fe atom in the protein.
- There are 8 different Fe-S proteins that function in mitochondrial electron transfer
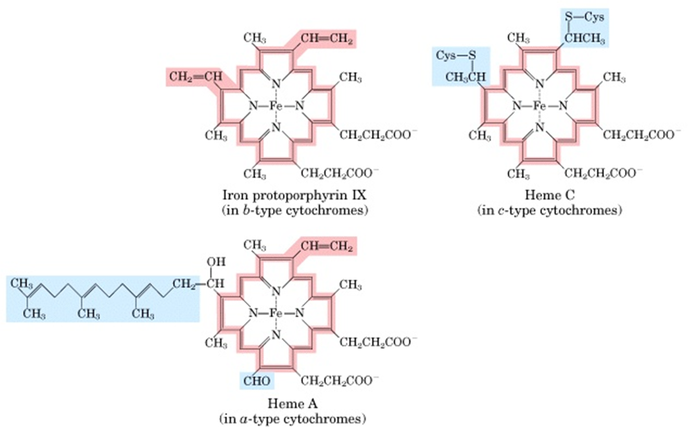
Cytochromes
- Cytochromes are proteins with a Fe-containing heme prosthetic group.
- They absorb light in visible range due to the heme group
- There are three classes of cytochromes depending of the type of heme group they have.
- Heme of cyt-a and b are tightly bound to protein (non-covalently) whereas that in c type cytochrome is covalently bound.
- Cyt-a and b are membrane proteins whereas cyt-c in mitochondria is a soluble protein associated with the outer surface of membrane by electroststic interaction.
Components of Mitochondrial Electron Transport Chain
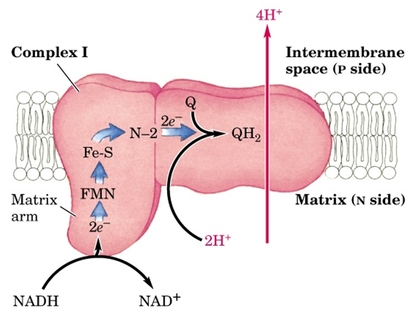
Complex-I : NADH-Coenzyme Q oxidoreductase:
- It is the largest protein complex in mitochondrial membrane containing
- 43 polypeptides and total approximate molecular weight 850kDa.
- It contains 6-7 iron sulfur clusters and 1 FMN as prosthetic group.
- It has binding site for Coenzyme Q which is a substrate for this complex
Complex II: Succinate dehydrogenase;
- Contain four different proteins
- One protein has a covalantly bound FAD prosthetic gp and an Fe-S centre with four Fe atoms. Another protein is also an iron-sulfur protein.
- Electrons pass from succinate to FAD and then through Fe-S centrs to Ubiquinone.
- Other mitochondrial dehydrogenases can pass electrons from different substrates to ubiquinone in the respiratory chain.
- During beta oxidation of fatty acids, acyl-CoA dehydrogenase catalyses the transfer of electron to FAD then to electron transferring flavoprotein (ETF) then to ETF:ubiquinone oxidoreductase. This enzyme passes electron to ubiquinone.
- Similarly, glycerol 3-phosphate dehydrogenase is a flavoprotein on the outer surface of inner mitochondrial membrane and transfer electron to Ubiquinone via FAD.
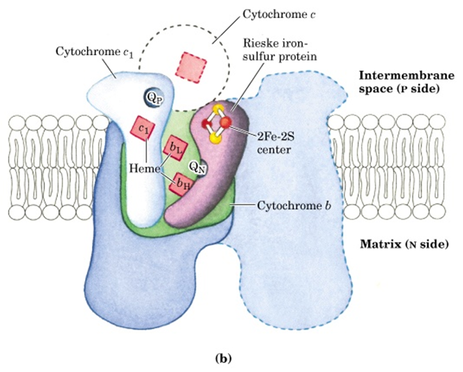
Complex III: Cytochrome bc1 complex or Ubiquinone:cytochrome c oxidoreductase complex.
- This complex is a dimer of two identical complexes: each containing 11 different subunits.
- Three subunits that form the functional core are cytochrome b with two hemes, Rieske iron sulfur centre and cytochrome c1. Electrons from QH2 are transferred via Fe-S centre to cyt.c1 and then to cyt c.
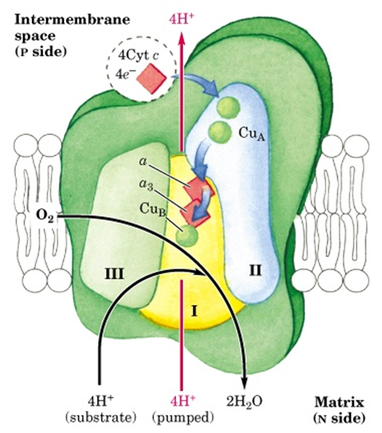
Complex IV: Cytochrome c oxidase: It catalyses the transfer of electron from cyt. C to oxygen leading to the reduction of oxygen to H2O.
- This complex has 13 subunits, total mol, mass 204 kDa). Three proteins critical for electron flow are: Subunit I, II and III.
- Subunit I contains two heme groups and a Cu ion (CuB).
- Subunit II contains two Cu atoms bonded by two sulfur atoms of cystiene. This binuclear centre and cyt c binding sites are located on towards the out side of the membrane.
- Subunit III: its role is not understood well, but it is essential for complex IV function.
- Electron flow takes place as indicated in the figure.